ICH Regulatory Compliance Navigator
Compliance Finder
Select your development focus to identify the relevant ICH guideline categories.
Guideline Result
Ensure you have selected a development phase.
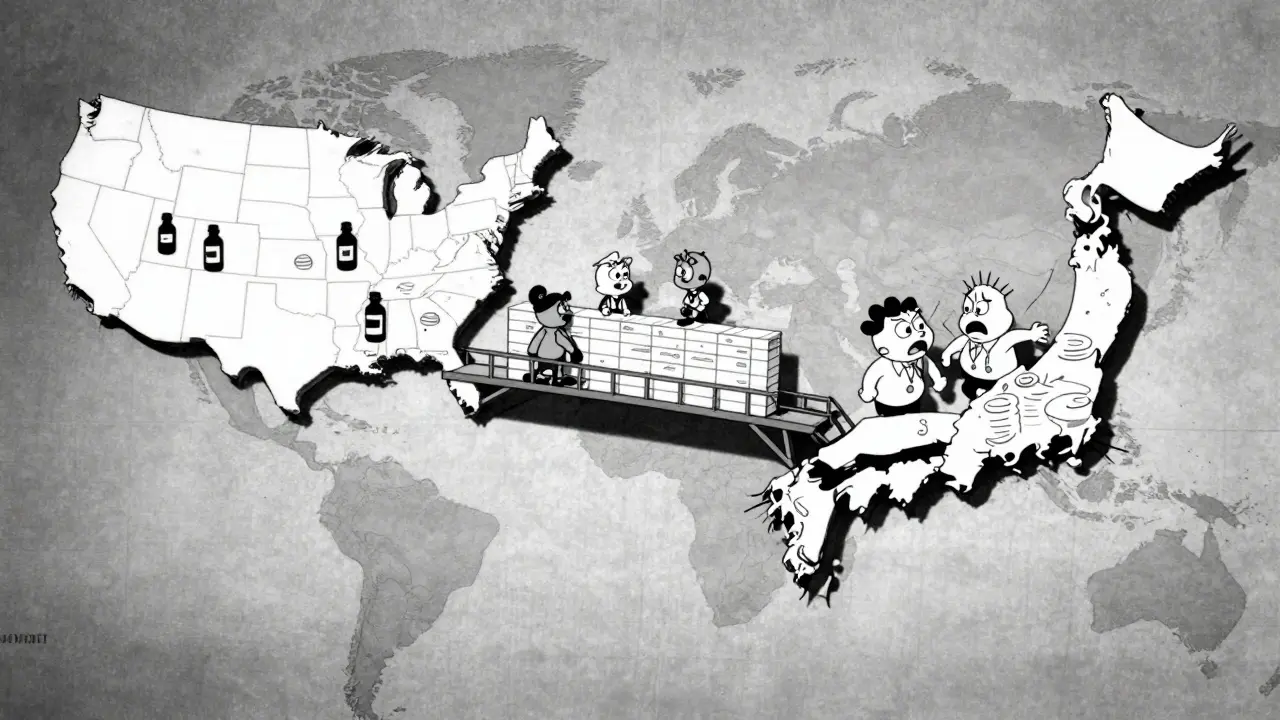
Imagine a scenario where a life-saving medicine is ready in the United States but sits waiting in Japan due to different testing requirements. Before 1990, this was a common nightmare for patients everywhere. Regulatory barriers meant duplication of work, delayed access, and unnecessary animal testing. Today, a global framework solves exactly that problem. We call this framework the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use.
The ICH isn’t just another bureaucratic committee. It is the bridge between the scientists developing medicines and the regulators approving them. For anyone working in the health sector, understanding this system is key to grasping how drugs reach patients safely and efficiently worldwide. As we move through 2026, looking back at the last few years of regulatory evolution gives us clear insight into where medication safety stands.
What Exactly Is the ICH?
To understand medication safety, you have to start with the organization managing it. International Council for Harmonisation is a unique global platform established in 1990 that brings together regulatory authorities and pharmaceutical industry representatives to develop and implement harmonized scientific and technical guidelines for drug development and registration. Also known as ICH, it operates under Swiss law since its legal restructuring in October 2015. This shift from a voluntary agreement to a non-profit legal entity changed everything. It expanded governance beyond the original founders and solidified its role in global health infrastructure.
Originally, this group was formed by the U.S., the European Union, and Japan. These three powers realized their differing standards were slowing down innovation. Now, the scope includes many more regions. The mission remains simple but profound: to ensure safe, effective, and high-quality medicines are developed in the most resource-efficient manner possible. Without this alignment, every country would demand separate tests for the same drug, wasting time and money that could go toward research.
How Guideline Development Works
You might wonder how a set of rules gets made when multiple governments are involved. It isn’t a quick vote. The ICH follows a rigorous five-step process designed to build true consensus among experts before any government commits to following the rule.
First, experts discuss a concept. Once there is general agreement on the science, they draft the guideline. This draft then goes to regulators for public feedback. Only after all concerns are addressed does it move to Step 4, where member jurisdictions officially adopt it. Finally, Step 5 involves implementation. Some guidelines take years to complete this journey. For instance, the ICH S1 guideline on carcinogenicity testing, adopted in November 2000, took significant time to mature. Patience here ensures that when a rule finally lands on a regulator’s desk, it is scientifically sound.
| Category Code | Focus Area | Key Purpose |
|---|---|---|
| Q (Quality) | Manufacturing Standards | Ensures consistent production quality |
| S (Safety) | Non-Clinical Testing | Evaluates toxicity and safety risks |
| E (Efficacy) | Clinical Trials | Proves the medicine works as intended |
| M (Multidisciplinary) | General Principles | Covers data reporting and bioequivalence |
Key Safety Standards and Guidelines
When people talk about medication safety, they often refer specifically to the 'S' category guidelines. These documents tell researchers exactly how to test a drug for potential harm before humans ever touch it. The ICH S1 guideline addresses carcinogenicity-checking if a drug causes cancer. Similarly, ICH S2 looks at genotoxicity, ensuring the drug won’t damage DNA. These aren’t suggestions; they are the global standard for preclinical risk assessment.
However, safety doesn’t stop at the lab. Clinical trials involving human subjects follow strict ethical codes defined by ICH E6 Good Clinical Practice. This guideline establishes the baseline for trial conduct worldwide. If a researcher in London follows ICH E6, the data is accepted by the FDA in the U.S. or PMDA in Japan. This cross-border acceptance is what makes global trials possible. Companies don’t have to run separate studies for every market.
Bioequivalence is another pillar. Recently, in June 2024, the UK’s Medicines and Healthcare products Regulatory Agency implemented ICH M13A. This guideline focuses on immediate-release solid oral dosage forms. In plain English, it ensures that generic versions of pills perform the same way as the brand-name originals. Patients get cheaper options without sacrificing effectiveness.

The Role of Regional Regulators
The ICH writes the rules, but local agencies enforce them. The relationship between the council and these agencies is crucial. U.S. Food and Drug Administration is a federal agency responsible for protecting public health by enforcing regulations on food, drugs, and cosmetics. The FDA treats ICH guidelines as official guidance. When the ICH publishes a finalized standard, the FDA generally implements it directly. This creates a de facto standard for anyone selling drugs in America.
In Europe, the situation involves the European Medicines Agency. The EMA maintains a directory of all implemented guidelines. While the process varies slightly, the goal remains the same: convergence. Post-Brexit changes added another layer of nuance. The UK’s MHRA transitioned from EU-associated membership to full ICH membership in May 2022. This allowed Britain to participate directly in the decision-making rather than just following European directives. It marked a significant expansion of the ICH ecosystem beyond the original tripartite foundation.
Industry players also sit at the table. Organizations like the International Federation of Pharmaceutical Manufacturers and Associations serve as Standing Observers. They represent company interests alongside government officials. This balance ensures that while safety is paramount, the rules remain practical enough for companies to actually implement. It prevents regulations from becoming theoretical ideals that nobody can execute.
Emerging Trends in Regulatory Science
We are living through a shift in how regulators view data. Historically, approval relied heavily on randomized controlled trials. But those trials capture only a snapshot of reality. Now, the focus is expanding to include real-world evidence. In June 2024, the ICH assembly adopted a reflection paper specifically on this topic. The aim is to harmonize terminology around real-world data and enable convergence on study protocols.
Why does this matter? Because once a drug hits the market, doctors track how it performs in diverse populations. Harmonizing how we collect and report that data means we spot safety signals faster. If a rare side effect appears in Germany, the format of the report allows the FDA to see it instantly. The integration of real-world evidence marks a maturing of the safety monitoring lifecycle.
There are challenges, naturally. Not every guideline applies perfectly to every therapeutic area. Gene therapies and AI-driven drug development are moving faster than traditional regulatory frameworks sometimes allow. Long-term viability depends on the organization continuing to adapt. Current frameworks cover chemicals and biologics well, but digital therapeutics pose new questions. The willingness to update procedures, however, suggests resilience.

Benefits for Patients and Society
It is easy to get lost in acronyms like ICH S1 or E6, but the bottom line is human lives. Expert analysis consistently highlights that international harmonization reduces patient burden. You avoid unnecessary duplication of clinical trials. A volunteer who participates in a trial in one country helps validate safety for patients globally.
Furthermore, the environmental and ethical benefit regarding animal testing cannot be overstated. By agreeing on methods upfront, the global community avoids repeating toxicology studies. The FDA explicitly notes that this leads to a reduction of unnecessary animal testing without compromising safety. This efficiency accelerates market entry too. Faster approvals mean patients get access to treatments sooner. Reduced administrative friction lowers costs, which theoretically translates to better pricing structures.
Practical Compliance for Professionals
If you are navigating this landscape, knowing where to look is essential. Regulatory authorities provide Question and Answer documents to clarify implementation. For example, the FDA published clarifications on ICH E3 regarding clinical study reports. Companies integrate these standards into Standard Operating Procedures. Compliance is mandatory in markets like the U.S., while other regions treat them as voluntary recommendations with very strong incentives to follow suit.
Training resources exist via the official ICH website. Learning how to read these guidelines helps professionals predict regulatory moves. Understanding the difference between Step 2 (Drafting) and Step 4 (Implementation) saves teams from preparing for rules that aren’t yet binding. Keeping an eye on the implementation status directories provided by the EMA or MHRA keeps you ahead of legislative changes.
The world of pharmacovigilance relies on trust. Trust that the rules are fair, scientific, and transparent. The ICH has proven to be the bedrock of that trust. As we look forward, the continued inclusion of new members and the adaptation to modern science will define the next chapter of medication safety.
Frequently Asked Questions
Are ICH guidelines legally binding laws?
Not inherently. ICH itself cannot pass laws. However, major regulators like the FDA and EMA adopt them into their own legal frameworks. For the US market, they function effectively as mandatory guidance documents that companies must follow to gain approval.
Who can join the ICH organization?
Membership is open to regulatory authorities and industry organizations. Originally founded by the EU, Japan, and USA, it expanded significantly. Countries or groups can apply for observer status or full membership, subject to consensus and fee payments.
How does ICH impact generic drug prices?
Harmonized bioequivalence standards (like ICH M13A) make it easier for generic manufacturers to enter different markets. Increased competition usually drives prices down while maintaining the same quality standards as brand-name drugs.
Can a country reject an ICH guideline?
Yes, implementation is ultimately voluntary for each jurisdiction. However, because major markets depend on trade, rejecting a global standard creates barriers to export and import. Most economies prefer alignment to facilitate trade and ensure safety.
What is the difference between ICH and WHO guidelines?
WHO focuses on global public health and accessibility in developing nations. ICH focuses on technical standards for registration and manufacturing primarily used in high-regulation markets. They often collaborate, but ICH standards are stricter on commercial product specifications.
Dan Stoof
March 31, 2026 AT 05:58This framework is absolutely fantastic for global health!
It really saves so many precious lives worldwide!
I love seeing how collaboration works so beautifully!
Every single patient deserves access to quality drugs!
We should celebrate every step forward in safety!
The future looks incredibly bright for medical science!
New technologies will thrive under these united rules!
People everywhere benefit from standardized testing methods!
Hope spreads when we remove bureaucratic barriers successfully!
Innovation flourishes when experts share their knowledge openly!
emma ruth rodriguez
April 1, 2026 AT 18:43The technical specifications are indeed quite rigorous.
Implementation protocols demand strict adherence to the text.
Deviations can compromise the entire validation process.
Professionals must maintain high competence levels.
Continued education is essential for compliance officers.
Regulatory clarity prevents costly errors during development.
Michael Kinkoph
April 3, 2026 AT 11:56Most people fail to grasp the depth of this issue.
True understanding requires intellectual rigor and discipline.
Bureaucracy is often a shield against incompetence elsewhere.
One must respect the complexity of regulatory science.
Casual observers miss the vital nuances completely.
Superficial praise ignores the immense effort required.
dPhanen DhrubRaaj
April 4, 2026 AT 08:04i think its cool how they work together like that ... and maybe its better for everyone eventually ... definitely less animals getting hurt ... thats nice ... we should keep supporting them ...
Adryan Brown
April 5, 2026 AT 18:17It really shows how much we need to work together.
Science does not respect borders when patients are suffering.
The old ways created so many delays for everyone involved.
We should look at the big picture instead of small details.
Everyone wants the same thing which is safe medicine.
That goal requires trust between different nations today.
Trust builds up when regulations align properly over time.
The environment benefits greatly from shared testing methods.
Animals do not suffer when we share the data sets.
Patients receive treatments sooner because of these efforts.
Cost savings help families afford necessary medical care.
Innovation moves faster without redundant bureaucratic hurdles.
Governments save money on their own administrative overhead.
Researchers focus more on discovery than compliance paperwork.
Future generations will thank us for this foundational work.
Angel Ahumada
April 7, 2026 AT 02:55the essence of harmony is finding truth beyond the boundaries of national ego ... we must transcend the materialistic drive for profit ... safety becomes a spiritual imperative rather than a legal one ...
Biraju Shah
April 8, 2026 AT 18:57Just implement the standards and move forward immediately.
Charles Rogers
April 9, 2026 AT 21:59Only the dedicated understand the value here.
Others see rules as obstacles to freedom.
Wisdom dictates following the established path.
Brian Yap
April 11, 2026 AT 19:40Pretty cool stuff honestly :)
Makes sense why things are moving fast now
Good for everyone i guess !
Jonathan Alexander
April 13, 2026 AT 09:29It is a magnificent tragedy how long it took!
Now we stand on the precipice of change!
History remembers the brave who harmonized!